A step-down (buck converter) is a DC-to-DC converter that reduces voltage when current is increased from its input supply to its load. These converters provide much better power efficiency than linear regulators. Its efficiency is frequently over 90%, which makes it very useful for different tasks like changing the main supply voltage of a computer. So, step-down converters normally have a minimum of two semiconductors like a transistor and a diode, while current buck converters often change the … [Read more...]
Voice-Controlled Home Automation using IFTTT and ESP8266
Home automation is no longer a luxury — it’s now affordable and achievable even for beginners. In this article, we’ll walk you through a voice-controlled home automation project using an ESP8266 Wi-Fi module and IFTTT, with no coding required. Whether you're automating a lamp or your entire home lighting, this voice-controlled home automation guide has you covered. Voice-Controlled Home Automation using IFTTT and ESP8266 The required components for basic lamp control include the follo … [Read more...]
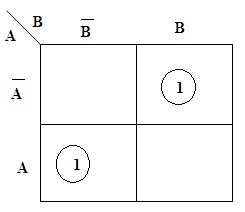
Half Subtractor : Circuit & Its Applications
Half Subtractor process can be analyzed from the decimal subtraction. The same concept can be utilized for binary numbers. The subtraction mechanism is used in many digital circuits. To perform addition the value of sum and in some cases, the carrier is also generated by using Binary Adders. But in this, the difference and the borrow area to be considered for subtraction. Half Subtractor Theory Half Subtractor is a kind of 'Combinational Circuit'. These are designed to achieve the difference … [Read more...]
What is Mesh Analysis : Methods & Its Uses
In network analysis, the basic and simple circuits can be analyzed by using basic techniques and laws like KVL, KCL, and ohm's law. In the case of complex circuits, these laws are not applicable and unreliable as it consists of controlled sources. Hence to find the variables in complex circuits, mesh, and nodal analysis techniques are used. By the use of mesh/loop and nodal analysis, we can determine the variables like current and voltages of the circuit very easily. This nodal analysis is used … [Read more...]
What is Peak Inverse Voltage & Its Importance
In electronics engineering, different kinds of materials are used to carry electricity. The materials like copper, aluminum, germanium Ge), silicon (si) are good conductors of electricity and have good conductivity. Such materials are called semiconductors, used in various electronic applications. These types of materials have a narrow forbidden gap of 1eV, sufficient to transfer the electrons from the valance band to the conduction band. When two types of materials like p-type and n-type are … [Read more...]
What is a Programming Language and Different Types
Nowadays, there are numerous programming languages are becoming more general and all-purpose, but these languages have their specialties, and each language has its own advantages and disadvantages. Usually, programming languages can be classified into a few types, however, these languages support multiple programming style. Every year there are a number of programming languages are implemented, but few languages are becoming very popular which may used by a professional programmer in their c … [Read more...]
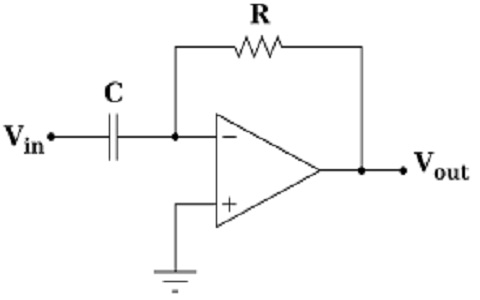
What is an Op Amp Differentiator : Circuit & Its Working
An operational amplifier or Op-Amp is a linear device that is used in ideal DC amplification, signal conditioning, filtering, and also in mathematical operations like addition, subtraction, integration, and differentiation. Op-amp is a voltage amplifying device which is having external feedback components such as capacitors and resistors between output and input terminals. The feedback components presenting in Op-Amp are useful to carry on the operation in an efficient way. An Op-Amp is a … [Read more...]
What is RMS Voltage : Theory & Its Equation
In electronics circuits, the signal is associated with an AC (Alternating current) and DC (Direct Current). The sinusoidal wave or sine wave represents the periodic AC signal that varies with time and fluctuates between positive and negative cycles. While DC signal/waveform represents the magnitude of voltage and current values easily and stable. But the magnitude of the current and voltage values of the AC signal should be calculated by using its instantaneous values, a peak value of the … [Read more...]
What is Snell’s Law : Definition & Its Derivation
Initially, Ptolemy a scientist from Alexandria discovered the relation that exists between refraction angles, but his invention failed for a minimal range of angles. In the scope of this Alhazen invented the law of refraction and finally, it was named Snell by the Persian researcher Ibn Sahl. After this many inventors worked upon this law to know the exact principle of this law. Finally, with the enhancement of various electromagnetic and optical concepts, Snell’s law was brought into the modern … [Read more...]
What is Hartley Oscillator : Working and Its Applications
An electronic oscillator in which the oscillator frequency is determined through a tuned circuit consisting of inductors and capacitors is called the Harley oscillator. The invention of the Harley oscillator circuit was done in 1915 by Ralph Hartley, an American engineer. This oscillator is a type of harmonic oscillator. The Hartley Oscillator produces the waves of radio frequencies therefore it is also referred to as radiofrequency oscillators. The tank circuit of the oscillator having a … [Read more...]
- 1
- 2
- 3
- …
- 12
- Next Page »