A crystal that has its conduction value in between conductor and insulator is termed as the semiconductor.It can be formed by the addition of impurities. It can be referred to as either p-type or n-type. Hence its conduction is based on the types and the amount of impurity added. These solids conduction value is proportional to the temperature value. As the temperature increases its conduction value increases as temperature degrades then the conduction of the semiconductor tends to decrease.
Semiconductor and its Types
These semiconductors are further classified into two basic types based on their purity and the doping value, For more detailed comparison between the two semiconductor types, refer to the article – Difference Between Intrinsic and Extrinsic Semiconductor
(1) Intrinsic Semiconductor
The semiconductor in its pure form is termed an intrinsic semiconductor. It is also related to its energy gaps for the silicon in its pure form the energy gap will be 1.1 electron volts and for germanium, it is 0.72 electron volts. In this type semiconductor at room temperature, the number of carriers and number of holes is equal to each other indicating the neutral condition.
(2) Extrinsic Semiconductor
Once the impurity is added to the semiconductor then its purity gets affected and also there is the increment in the charge carriers. These types of semiconductors are known as extrinsic semiconductors. Further, this is classified into two types’ p-type and n-type. In these types, the numbers of electrons are more in n-type whereas the number of holes in p-type.
What is a P-Type Semiconductor?
If the intrinsic semiconductor is doped with an electron acceptor in order to make it as a p-type semiconductor. The electron acceptor is responsible for the formation of a hole by accepting an electron from the lattice.
As a result, majority carriers in the p-type semiconductor formed are holes. In this way, a p-type semiconductor is defined based on its electron acceptor capability.
Formation of P-type Semiconductor
In order to form a p-type semiconductor the basic step is to dope intrinsic semiconductor with trivalent impurity. In this type, the valence shell consists of three electrons requires further one more electron.
This is possible by sharing the electron. As it is accepting electrons it is generally referred to as acceptor. The acceptor impurities are Boron, indium, gallium. Once these are added to either silicon or germanium p-type semiconductors are formed.
Let us take boron as the trivalent impurity so that we can add it to the silicon in order to make it extrinsic. Everything here is completely based on the concept of sharing.
The covalent bond concept is preferred during accepting the electrons. Boron atomic number is five. Based on its valence shell electronic configuration concept the numbers of valence electrons present in boron are three.
Hence each valence electron forms a covalent bond with the neighboring silicon atoms. The number of bonds formed with a number of silicon atoms is based on the number of valence electrons present.
It is evident that to boron there are four neighbouring silicon atoms as per that there must be four electrons but there are only three valence electrons present. Then the absence of fourth electron or the vacancy of the electron is termed as a hole.
It indicates that one electron is accepted by the boron atom. The vacancy or the need of electron can be fulfilled. The number of vacancies and the acceptance of electrons are proportional to each other.
Now it is clear that boron comes under trivalent impurity because it is responsible for the formation of holes in the semiconductor. Therefore doping pure form of silicon with boron leads to the formation of p-type semiconductor.
Please refer to this link for Semiconductor Device MCQs
Example of P-Type Semiconductor
Trivalent impurities such as boron or gallium are commonly used in silicon as doping impurity. Then silicon doped with boron or gallium is a perfect example for a p-type semiconductor.
Whether silicon is doped with gallium or indium the process is also can be represented by utilizing the same concept of boron and silicon.
Silicon Doped with Boron
In this way, the semiconductors that are rich in holes as there carriers formed by the trivalent impurities comes under the list of p-type semiconductors.
Whether silicon or germanium if they are added with a trivalent impurity that belongs to p-type of the semiconductor.
Conduction in P-Type Semiconductor
When the external supply of voltage is given to the p-type semiconductor there majority carriers present in valence band tends to move towards the negative terminal of the supply and the minority carriers that are electrons present in the conduction band move towards the positive terminal.
Conduction in P-Type Semiconductor
However, the concentration of electrons is less in the conduction band and the majority of holes are present in the valence band.
Hence the current in the p-type is because of majority carriers in valence band a little amount of current is in conduction band because of few electrons that are their minority carriers.
Energy Diagram of P-Type Semiconductor
As it is doped with trivalent impurity there are a huge number of holes formed in the p-type. Hence it has a majority concentration of holes and minority concentration of electrons in it.
P-type because of majority a-holes it referred to as a positive type. As it is a p-type semiconductor the Fermi level is present near to the valence band rather than conduction band.
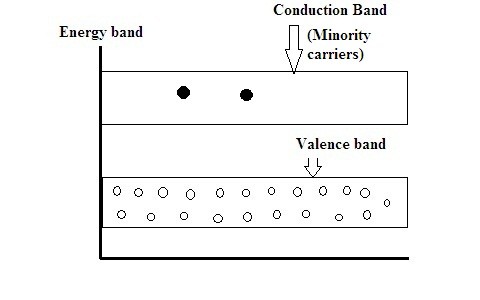
Energy Band Diagram of P-Type Semiconductor
Once the impurity is inserted in the pure semiconductor numerous amounts of holes are formed in the valence band. There are thermal excitation’s also in the semiconductors because of this same amount of electrons are also present in the conduction band.
As always majority wins, more amount of hole compared to electrons makes p- types majority carriers as holes.
As the energy band diagram suggests there is less number of electrons in conduction band in p-type compare to that of n-type making the state of conductivity of n-type double than that of p-type.
The reason behind this is the excitation’s will release a tremendous amount of energy and also enhances the flow of current making to mold it purposefully based on requirements.
As the doping concept introduced in extrinsic and made it p-type and n-type. The formation of these types made changes in the development of modern electronics.
Please refer to this link to know more about Semiconductor Theory MCQs
Please refer to this link to know more about Semiconductor Diode
For example, it can be LASER, LED or a BJT everything is interlinked to each other. In what ways do you think p-type is preferred over n-type?